Introduction
Autosomal recessive spastic ataxia of Charlevoix-Saguenay (ARSACS) is a rare neurodegenerative disorder predominantly affecting the nervous system.
The prevalence of ARSACS has been reported as 1/1932 live births in the Charlevoix-Saguenay region of Quebec, Canada, where it was first identified [1]. Such high prevalence is attributed to a founder effect in this geographically isolated population. Outside of Quebec, however, ARSACS is considered exceedingly rare, and the worldwide prevalence remains unknown. Nevertheless, recent studies have identified ARSACS cases in various regions, including Europe [2] and East Asia [3], suggesting that its global distribution may be broader than initially recognized.
ARSACS is characterized by early-onset spasticity, ataxia, and peripheral neuropathy [4]. Typically presenting in infancy or early childhood, the disease leads to progressive motor dysfunction, coordination issues, and muscle weakness. Neurological features of ARSACS frequently include cerebellar atrophy and microcephaly, contributing to the gradual decline in motor control and overall function [5, 6].
Although respiratory failure is not a primary feature of ARSACS, the neuromuscular weakness associated with the disorder can impair respiratory muscle function over time, potentially leading to respiratory complications. The management of such complications in a rare and progressively debilitating condition like ARSACS presents significant challenges.
Non-invasive ventilation (NIV) is a recognized therapeutic approach for managing respiratory failure in various neurodegenerative and neuromuscular disorders [7]. By providing ventilatory support without the need for invasive procedures, NIV can improve gas exchange, ease the burden on respiratory muscles, and enhance patient quality of life [8].
This case report presents a patient with ARSACS who developed respiratory failure and was successfully managed using NIV. This case highlights the role of non-invasive respiratory support in rare neurodegenerative conditions and underscores the importance of early intervention for respiratory complications in patients with ARSACS.
Case Report
A 52-year-old male was admitted to the Pulmonary Rehabilitation Center at Gangnam Severance Hospital due to complaints of chronic fatigue, headaches, and shortness of breath, particularly during speech and with exertion. He also reported difficulty clearing his airway due to excessive sputum production and a reduced ability to cough.
The patient has a long history of progressive neurological decline, which began in his early childhood. He experienced delayed growth and developed gait disturbances by elementary school. By high school, he frequently fell down due to weakness and by his sophomore year, independent walking was no longer possible. Since his 30s, the patient has been wheelchair-dependent and his condition further exacerbated, showing symptoms of dysarthria. In his 40s, the patient became bed-ridden.
Neurological Workup
Neurological assessment revealed a borderline cognitive function with a Korean-Mini Mental State Examination (K-MMSE) score of 26, which is slightly below the normal range. This assessment was performed to rule out severe cognitive impairment which is typically associated with other forms of neuromuscular diseases. The patient exhibited pronounced cerebellar dysfunction, including dysmetria in the finger-to-nose test, dysdiadochokinesia, and intentional tremor. Resting tremor and postural tremor were absent in his case. Motor strength was reduced in both the upper and lower limbs, with a Medical Research Council grade of P+/T in the lower extremities and F/F in the upper extremities. Spasticity measured using Modified Ashworth Scale (MAS) was graded as G2 in the lower extremities; spasticity was not observed in the upper extremities. Proprioception, assessed using the joint reposition test, was found to be impaired bilaterally below the ankles, while light touch and pain sensations were both preserved. The patient exhibited severe gait disturbances and coordination difficulties, with a Berg Balance Scale (BBS) score of 2/56 upon admission. The patient was unable to walk or stand independently, maintaining balance only while sitting, and required assistance for all activities of daily living.
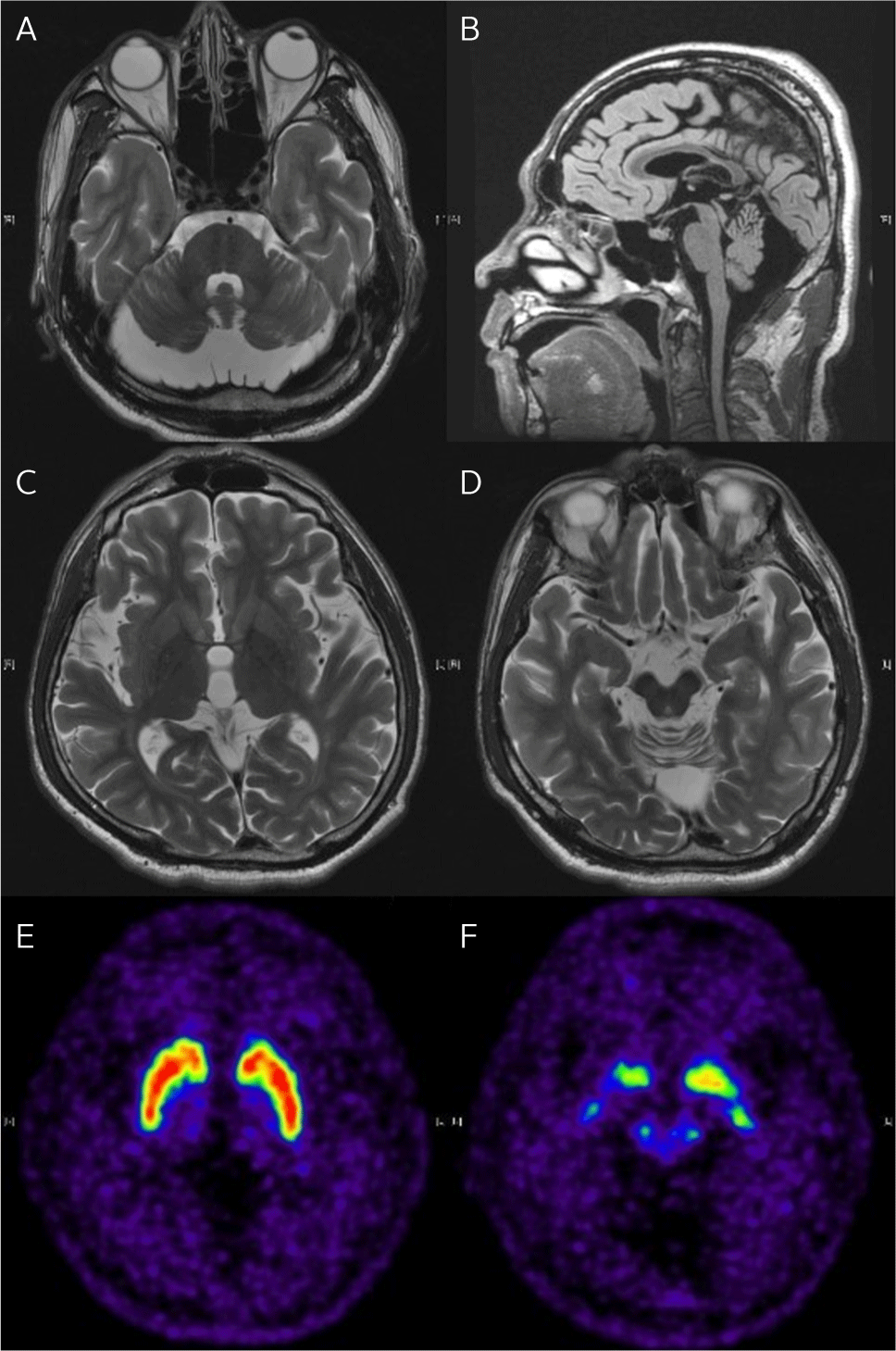
As shown in Fig. 1, brain magnetic resonance imaging (MRI) showed cerebellar atrophy, particularly in the vermis and superior cerebellum, without significant brainstem involvement. Enlarged cerebrospinal fluid spaces in the posterior cranial fossa were observed without fourth ventricle enlargement, suggesting the presence of mega cisterna magna. No other abnormal brain findings, such as ischemic lesions or masses, were observed. Additionally, [18F] N-3-fluoropropyl-2-beta-carbomethoxy-3-beta-(4-iodophenyl) nortropane (F-18 FPCIT PET/CT) was normal. The patient exhibited retinal nerve fiber layer swelling, a characteristic feature of ARSACS.
Whole genome sequencing revealed a heterozygous stop-gained mutation in the SACS gene (c.7272G>T, C2424), which is consistent with ARSACS. In addition, a damaging heterozygous missense mutation in the PRX gene (c.3239C>T, R1080H) was found, associated with Charcot-Marie-Tooth disease and Dejerine-Sottas disease.
Based on the clinical findings and genetic testing, the patient was diagnosed with ARSACS.
Respiratory Evaluation
Upon admission, the patient's pulmonary function was assessed. Pulmonary function tests revealed severe restrictive lung disease. His sitting vital capacity (VC) was 1140 mL (21.2% of predicted), and his supine VC was 1050 mL (19.6% of predicted). Peak cough flow was measured at 110 L/min, indicating significantly reduced coughing capacity. A chest CT scan was performed, and no abnormalities in the lung parenchyma, such as pneumonia or fibrosis, were observed. Arterial blood gas analysis showed a PaCO2 level of 48.2 mmHg, suggesting hypercapnia, and an oxygen saturation of 98.0%.
Overnight transcutaneous CO2 monitoring confirmed hypercapnia, with maximum CO2 levels of 51.9 mmHg and 54.4 mmHg on two consecutive nights with mean CO2 levels around 47-50 mmHg, indicating chronic CO2 retention and impaired ventilation. These findings were consistent with the patient's reported symptoms of fatigue, headaches, and mental cloudiness, all suggestive of hypercapnic respiratory failure.
NIV was initiated to manage the patient’s chronic respiratory insufficiency. Following our protocol [9, 10], NIV settings were adjusted for optimal ventilatory support, using BiPAP mode with an inspiratory positive airway pressure of 22 cmH2O, an expiratory positive airway pressure of 0 cmH2O, and a respiratory rate of 15 breaths per minute. The non-invasive ventilatory support was delivered via a Wisp mask with a chin strap and was applied overnight.
Over time, the patient experienced significant improvement in various symptoms, including excessive sputum production and shortness of breath. We followed the established protocol of assessing the patient’s condition every 3 to 6 months in the outpatient clinic and performing overnight transcutaneous ventilator monitoring during annual hospital admissions. At the five-year follow-up, the patient reported no significant respiratory symptoms, showed no symptoms of respiratory insufficiency. However, there was a notable decline in his respiratory function. Forced vital capacity (FVC) in the sitting position decreased from 1,050 mL (19.6% predicted) to 540 mL (10.4% predicted), and peak cough flow dropped from 110 L/min to 90 L/min. Muscle strength, as assessed by the Medical Research Council scale, further deteriorated, with lower limb strength declining from P+/F to T/T. Functionally, the patient’s condition worsened to the extent that he could no longer maintain independent sitting balance.
Despite the decline in the overall respiratory and functional capacity, hypercapnia improved with consistent use of non-invasive ventilation (NIV). At the five-year follow-up, overnight transcutaneous CO2 monitoring demonstrated stable ventilation, with maximum CO2 levels of 43.2 mmHg and 39.1 mmHg, and mean levels of 36.6 mmHg and 33.2 mmHg on two consecutive nights. These findings underline the effectiveness of overnight NIV in managing this patient’s ventilatory insufficiency.
Discussion
This is the first reported case of respiratory insufficiency in a patient with ARSACS. ARSACS is primarily characterized by neurological symptoms such as cerebellar atrophy, spasticity, and peripheral neuropathy. It is also known to cause muscle weakness in the limbs. Previous literature delineates progressive decline in muscle strength followed by muscle atrophy that affects both the upper and lower limbs [2]. These muscular weaknesses are attributed to pyramidal manifestations and peripheral neuropathy [11].
This case suggests that patients diagnosed with ARSACS could not only exhibit muscle weakness of limbs but can also demonstrate weakness of the respiratory muscles. Over time, respiratory muscle weakness may lead to restrictive lung disease and, ultimately, to ventilatory failure. Notably, we have previously reported a case of a patient diagnosed with Freeman-Burian syndrome, a condition that is primarily associated with distal muscle weakness, who showed symptoms of respiratory muscle weakness [12]. Similarly, through this case, we were able to discover that patients with ARSACS could also experience respiratory muscle weakness. However, such respiratory decline could be undermined due to more noticeable symptoms such as reduced motility, potentially leading to the neglect of respiratory insufficiency. Thus, case studies are needed to confirm respiratory insufficiency in ARSACS, and, if feasible, histological examinations of respiratory muscles should be conducted to investigate any structural or functional changes in ARSACS patients.
The successful use of NIV in this patient underscores its potential as a life-saving intervention in ARSACS patients who develop respiratory complications. Early intervention with NIV proved effective in this case, alleviating the patient's symptoms of fatigue, headaches, and mental cloudiness. NIV has been well-established as a non-invasive method to manage chronic respiratory failure in neuromuscular disorders [13]. By providing ventilatory support, NIV reduces the work of breathing, improves gas exchange, and resolves hypercapnia. The success of NIV in this patient underscores its role as an essential tool in managing ARSACS patients who develop respiratory complications. Importantly, initiating NIV early can prevent further deterioration and improve the patient's quality of life, as demonstrated by the resolution of the patient’s CO2 retention and symptom relief.
Given that this is the first documented case of respiratory failure in ARSACS, it emphasizes the need for regular ventilatory monitoring, even in patients where respiratory complications are not imminent [10]. Early detection through pulmonary function tests and CO2 monitoring can lead to timely intervention, improving outcomes and quality of life. While this case demonstrates the benefits of NIV in managing respiratory failure in ARSACS, long-term follow-up is critical to monitor disease progression and adjust treatment strategies accordingly. In this case, five years of follow-up showed stable respiratory function with the continued use of NIV. This reinforces the importance of ongoing respiratory assessments and timely adjustments in ventilatory support to prevent further respiratory decline.
In conclusion, this case demonstrates that patients with ARSACS may develop significant respiratory complications as a result of neuromuscular weakness. NIV offers a safe and effective method to manage chronic respiratory failure, which alleviates respiratory complications, and enhances the quality of life in these patients. Further research is warranted to better understand the respiratory aspects of ARSACS and the role of early intervention in improving long-term outcomes.